Recombinant Human BMP-4 GMP Protein, CF GMP Best Seller
R&D Systems, part of Bio-Techne | Catalog # 314E-GMP

Key Product Details
Source
Accession #
Structure / Form
Conjugate
Applications
Product Specifications
Source
Lys303-Arg408
Produced in an animal-free laboratory. Manufactured and tested under cGMP guidelines
Purity
Endotoxin Level
N-terminal Sequence Analysis
Predicted Molecular Mass
SDS-PAGE
Activity
The ED50 for this effect is 3-21 ng/mL.
Host Cell Protein
Mycoplasma
Host Cell DNA
Scientific Data Images for Recombinant Human BMP-4 GMP Protein, CF
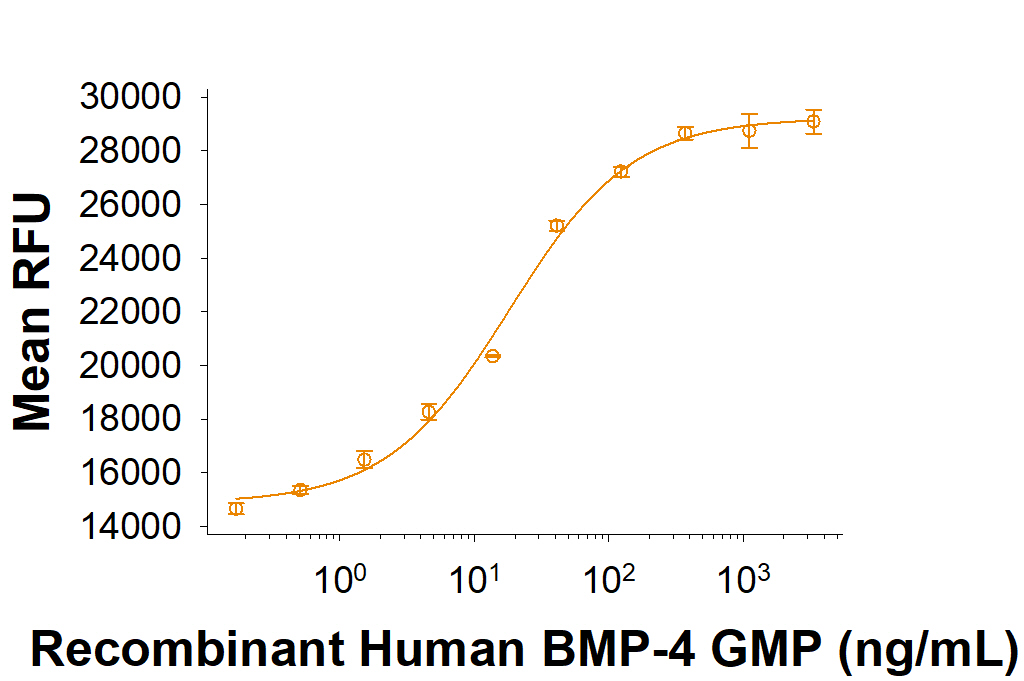
Equivalent Bioactivity of GMP and Animal-Free grades of Recombinant Human BMP-4
Equivalent bioactivity of GMP (Catalog # 314E-GMP) and Animal-Free (AFL314E) grades of Recombinant Human BMP-4 as measured by its ability to induce BMP responsive SEAP reporter activity in HEK293 human embryonic kidney cells (orange and green respectively).Formulation, Preparation and Storage
314E-GMP
| Formulation | Lyophilized from a 0.2 μm filtered solution in Acetonitrile and TFA. |
| Reconstitution | Reconstitute at 50-200 μg/mL in sterile 4 mM HCl. |
| Shipping | The product is shipped with polar packs. Upon receipt, store it immediately at the temperature recommended below. |
| Stability & Storage | Use a manual defrost freezer and avoid repeated freeze-thaw cycles.
|
Background: BMP-4
References
- Zhang, P. et al. (2008) Blood 111:1933.
- Gambaro, K. et al. (2006) Cell Death Differ. 13:1075.
- Simic, P. and S. Vukicevic (2005) Cytokine Growth Factor Rev. 16:299.
- Sadlon, T.J. et al. (2004) Stem Cells 22:457.
- Frank, D.B. et al. (2005) Circ. Res. 97:496.
- Lee, J-H. et al. (2014) Cell. 156:440.
- Tang, QQ. et Lane MD. (2012) Annu Rev. Biochem. 81:715.
- Urist, MR. (1965). Science. 150:893.
- Bandyopadhyay, A. et al. (2006) Plos Genetics. 2:e216.
- Wozney, J. et al. (1988) Science 242:1528.
- Cui, Y. et al. (1998) EMBO J. 17:4735.
- Cui, Y. et al. (2001) Genes Dev. 15:2797.
- Aono, A. et al. (1995) Biochem. Biophys. Res. Commun. 210:670.
- Nishimatsu, S. and G.H. Thomsen (1998) Mech. Dev. 74:75.
- Chen, D. et al. (2004) Growth Factors 22:233.
- Lavery, K. et al. (2008) J. Biol. Chem. April 24 epub.
- Rosen, V. (2006) Ann. N.Y. Acad. Sci. 1068:19.
- Jones, C.M. and J.C. Smith (1998) Dev. Biol. 194:12.
- Takada, T. et al. (2003) J. Biol. Chem. 278:43229.
Long Name
Alternate Names
Gene Symbol
UniProt
Additional BMP-4 Products
Product Documents for Recombinant Human BMP-4 GMP Protein, CF
Manufacturing Specifications
GMP ProteinsR&D Systems, a Bio-Techne Brand's GMP proteins are produced according to relevant sections of the following documents: USP Chapter 1043, Ancillary Materials for Cell, Gene and Tissue-Engineered Products and Eu. Ph. 5.2.12, Raw Materials of Biological Origin for the Production of Cell-based and Gene Therapy Medicinal Products.
R&D Systems' quality focus includes:
- Manufactured and tested under an ISO 9001:2015 and ISO 13485:2016 certified quality system
- Documented processes and QA control of documentation and process changes
- Personnel training programs
- Raw material testing and vendor qualification/monitoring
- Fully validated equipment, processes and test methods
- Equipment calibration schedules using a computerized calibration program
- Facility maintenance, safety programs and pest control
- Material review process for variances
- Monitoring of stability over product shelf-life
R&D Systems strives to provide our customers with the analytical characteristics of each product so that customers may determine whether our products are appropriate for their research. The Certificate of Analysis provided contains the following lot specific information:
- N-terminal amino acid analysis, SDS-PAGE analysis, and endotoxin level (as determined by LAL assay) performed on each bulk QC lot, not on individual bottlings of each QC lot
- Post-bottling lot-specific bioassay results (compliance with an established range) and results of microbial testing according to USP <71>
- Host Cell Protein testing performed by ELISA
- Mycoplasma testing by ribosomal RNA hybridization assay
Additional testing and documentation requested by the customer can be arranged at an additional cost.
Production records and facilities are available for examination by appropriate personnel on-site at R&D Systems in Minneapolis, Minnesota USA.
R&D Systems sells GMP grade products for preclinical or clinical ex vivo cell therapy applications. They are not for in vivo use. Please read the following End User Terms prior to using this product.
Animal-Free Manufacturing Conditions
Our dedicated controlled-access animal-free laboratories ensure that at no point in production are the products exposed to potential contamination by animal components or byproducts. Every stage of manufacturing is conducted in compliance with R&D Systems' stringent Standard Operating Procedures (SOPs). Production and purification procedures use equipment and media that are confirmed animal-free.
Production
- All molecular biology procedures use animal-free media and dedicated labware.
- Dedicated fermentors are utilized in committed animal-free areas.
- Protein purification columns are animal-free.
- Bulk proteins are filtered using animal-free filters.
- Purified proteins are stored in animal-free containers in a dedicated cold storage room.
Quality Assurance
- Low Endotoxin Level.
- No impairment of biological activity.
- High quality product obtained under stringent conditions.
Product Specific Notices for Recombinant Human BMP-4 GMP Protein, CF
END USER TERMS OF USE OF PRODUCT
The following terms are offered to you upon your acceptance of these End User Terms of Use of Product. By using this product, you indicate your acknowledgment and agreement to these End User Terms of Use of Product. If you do not agree to be bound by and comply with all of the provisions of these End User Terms of Use of Product, you should contact your supplier of the product and make arrangements to return the product.
We suggest you print and retain a copy of these End User Terms of Use of Product for your records.
The End User is aware that R&D Systems, Inc. sells GMP products for preclinical or clinical ex vivo use and not for in vivo use. The End User further agrees, as a condition of the sale of R&D Systems' GMP products that: a) the End User will not use this GMP Product in any procedure wherein the product may be directly or indirectly administered to humans, unless the End User has obtained, or prior to their use will have obtained, an Investigational New Drug (IND) exemption from the FDA and will use the product only in accordance with the protocols of such IND and of the Institutional Review Board overseeing the proposed research, or b) the End User will use the products outside of the United States in accordance with the protocols of research approved by the Institutional Review Board or authorized ethics committee and regulatory agencies to which the End User is subject to in their territory.
R&D Systems, Inc. has the right, at its sole discretion, to modify, add or remove any terms or conditions of these End User Terms of Use without notice or liability to you. Any changes to these End User Terms of Use are effective immediately following the printing of such changes on this product insert. The most recent version of these End User Terms of Use of Product may be found at: RnDSystems.com/Legal.
You agree to review these End User Terms of Use of Product to ensure any subsequent use by you of R&D Systems' GMP Products following changes to these End User Terms of Use of Product constitutes your acceptance of all such changes.
TERMS AND CONDITIONS
The following limitation applies to R&D Systems' warranty and liability for damages: All products are warranted to meet R&D Systems' published specifications when used under normal laboratory conditions.
R&D SYSTEMS DOES NOT MAKE ANY OTHER WARRANTY OR REPRESENTATION WHATSOEVER, WHETHER EXPRESS OR IMPLIED, WITH RESPECT TO ITS PRODUCTS. IN PARTICULAR, R&D SYSTEMS DOES NOT MAKE ANY WARRANTY OF SUITABILITY, NONINFRINGEMENT, MERCHANTABILITY OR FITNESS FOR ANY PARTICULAR PURPOSE.
NOTWITHSTANDING ANY OTHER PROVISIONS OF THESE TERMS AND/OR ANY OTHER AGREEMENT BETWEEN R&D SYSTEMS AND PURCHASER FOR THE PURCHASE OF THE PRODUCTS, R&D SYSTEMS' TOTAL LIABILITY TO PURCHASER ARISING FROM OR IN RELATION TO THESE TERMS, AN AGREEMENT BETWEEN THE PARTIES OR THE PRODUCTS, WHETHER ARISING IN CONTRACT, TORT OR OTHERWISE SHALL BE LIMITED TO THE TOTAL AMOUNT PAID BY PURCHASER TO R&D SYSTEMS FOR THE APPLICABLE PRODUCTS. IN NO EVENT WILL R&D SYSTEMS BE LIABLE FOR THE COST OF PROCUREMENT OF SUBSTITUTE GOODS.
Full details of R&D Systems' Terms and Conditions of Sale can be found online at: RnDSystems.com/Legal.
For preclinical, or clinical ex vivo use